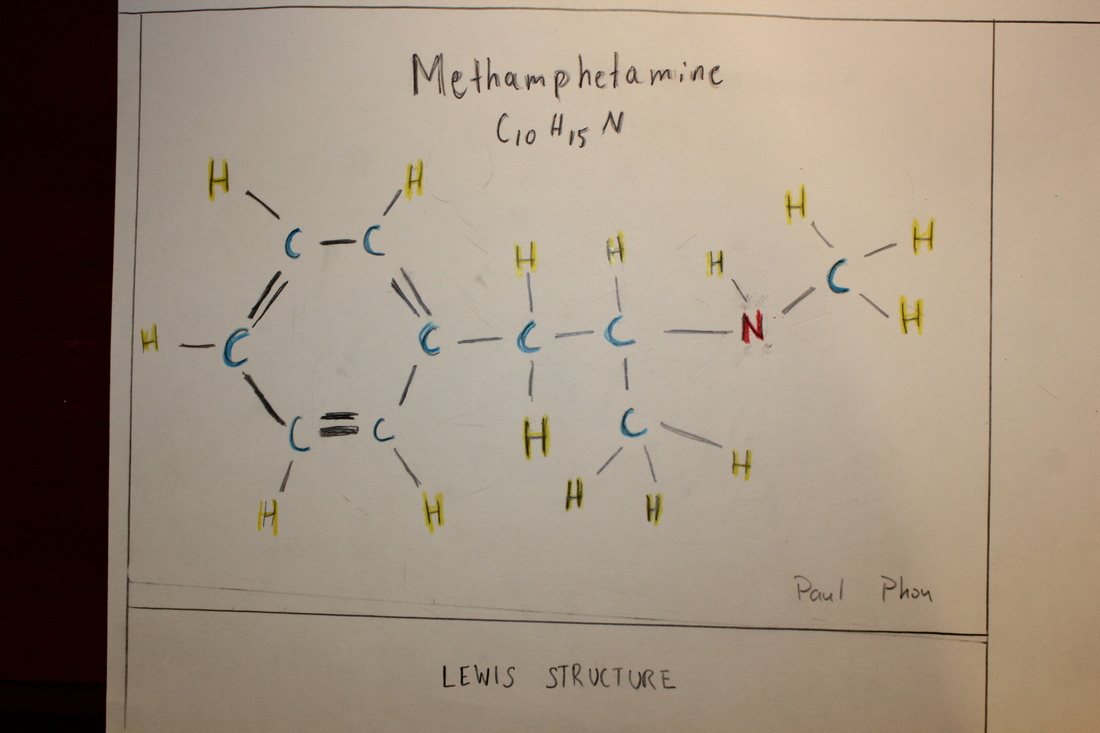
Lewis Structure
COLOR KEY
Blue = Carbon
Yellow = Hydrogen
Red = Nitrogen
Single Line = Single Bond
Double Line = Double Bond
Above is the Lewis structure for methamphetamine, C10H15N.
There is no VSPER form for this molecule, however, it can be considered to be a cyclical-chain molecule. The 6 carbon ring has a chain that extends from it.
The CH3 elements of the molecule are AX4 and tetrahedrons. The bond angle they form between the C and the H is 109.5 degrees. The angles between the hydrogens is 120 degrees.
The 6-carbon ring has bond angles of 120 degrees.
The nature of the bond between carbon and hydrogen is very covalent. (2.5-2.2=0.3)
The nature of the bond between nitrogen and carbon is very covalent. (3.0-2.5=0.5)
The nature of the bond between nitrogen and hydrogen is moderately covalent. (3.0-2.2=0.8)
The nature of the bond between carbon and carbon is completely covalent (2.5-2.5 = 0)
Blue = Carbon
Yellow = Hydrogen
Red = Nitrogen
Single Line = Single Bond
Double Line = Double Bond
Above is the Lewis structure for methamphetamine, C10H15N.
There is no VSPER form for this molecule, however, it can be considered to be a cyclical-chain molecule. The 6 carbon ring has a chain that extends from it.
The CH3 elements of the molecule are AX4 and tetrahedrons. The bond angle they form between the C and the H is 109.5 degrees. The angles between the hydrogens is 120 degrees.
The 6-carbon ring has bond angles of 120 degrees.
The nature of the bond between carbon and hydrogen is very covalent. (2.5-2.2=0.3)
The nature of the bond between nitrogen and carbon is very covalent. (3.0-2.5=0.5)
The nature of the bond between nitrogen and hydrogen is moderately covalent. (3.0-2.2=0.8)
The nature of the bond between carbon and carbon is completely covalent (2.5-2.5 = 0)